There are three methods of metalworking: hot working, cold working, and extruding. The method used depends on the metal involved and the part required, although in some instances both hot and cold working methods may be used to make a single part.
The rolled shape is called a bloom when its section dimensions are 6 inches × 6 inches or larger and square. The section is called a billet when it is square and less than 6 inches × 6 inches. Rectangular sections, which have a width greater than twice their thickness, are called slabs. The slab is the intermediate shape from which sheets are rolled.
Blooms, billets, or slabs are heated above the critical range and rolled into a variety of shapes of uniform cross section. Common rolled shapes are sheet, bar, channel, angle, and I-beam. Hot-rolled material is frequently finished by cold rolling or drawing to obtain accurate finish dimensions and a bright, smooth surface.
Complicated sections, which cannot be rolled, or sections of which only a small quantity is required, are usually forged. Forging of steel is a mechanical working at temperatures above the critical range to shape the metal as desired. Forging is done either by pressing or hammering the heated steel until the desired shape is obtained.
Pressing is used when the parts to be forged are large and heavy; this process also replaces hammering where high-grade steel is required. Since a press is slow acting, its force is uniformly transmitted to the center of the section, thus affecting the interior grain structure, as well as the exterior to give the best possible structure throughout.
Hammering can be used only on relatively small pieces. Since hammering transmits its force almost instantly, its effect is limited to a small depth. Thus, it is necessary to use a very heavy hammer or to subject the part to repeated blows to ensure complete working of the section. If the force applied is too weak to reach the center, the finished forged surface is concave. If the center was properly worked, the surface is convex or bulged. The advantage of hammering is that the operator has control over both the amount of pressure applied and the finishing temperature and can produce small parts of the highest grade. This type of forging is usually referred to as smith forging. It is used extensively where only a small number of parts are needed. Considerable machining time and material are saved when a part is smith forged to approximately the finished shape.
Steel is often harder than necessary and too brittle for most practical uses when put under severe internal strain. To relieve such strain and reduce brittleness, it is tempered after being hardened. This consists of heating the steel in a furnace to a specified temperature and then cooling it in air, oil, water, or a special solution. Temper condition refers to the condition of metal or metal alloys with respect to hardness or toughness. Rolling, hammering, or bending these alloys, or heat treating and aging them, causes them to become tougher and harder. At times, these alloys become too hard for forming and must be re-heat treated or annealed.
Metals are annealed to relieve internal stresses, soften the metal, make it more ductile, and refine the grain structure. Annealing consists of heating the metal to a prescribed temperature, holding it there for a specified length of time, and then cooling the metal back to room temperature. To produce maximum softness, the metal must be cooled very slowly. Some metals must be furnace cooled; others may be cooled in air.
Normalizing applies to iron base metals only. Normalizing consists of heating the part to the proper temperature, holding it at that temperature until it is uniformly heated, and then cooling it in still air. Normalizing is used to relieve stresses in metals.
Strength, weight, and reliability are three factors that determine the requirements to be met by any material used in airframe construction and repair. Airframes must be strong and yet as lightweight as possible. There are very definite limits to which increases in strength can be accompanied by increases in weight. An airframe so heavy that it could not support a few hundred pounds of additional weight would be of little use.
All metals, in addition to having a good strength-toweight ratio, must be thoroughly reliable, thus minimizing the possibility of dangerous and unexpected failures. In addition to these general properties, the material selected for a definite application must possess specific qualities suitable for the purpose.
The material must possess the strength required by the dimensions, weight, and use. The five basic stresses that metals may be required to withstand are tension, compression, shear, bending, and torsion.
The tensile strength of a material is its resistance to a force, which tends to pull it apart. Tensile strength is measured in pounds per square inch (psi) and is calculated by dividing the load in pounds required to pull the material apart by its cross-sectional area in square inches.
The compression strength of a material is its resistance to a crushing force, which is the opposite of tensile strength. Compression strength is also measured in psi. When a piece of metal is cut, the material is subjected, as it comes in contact with the cutting edge, to a force known as shear. Shear is the tendency on the part of parallel members to slide in opposite directions. It is like placing a cord or thread between the blades of a pair of scissors (shears). The shear strength is the shear force in psi at which a material fails. It is the load divided by the shear area.
Bending can be described as the deflection or curving of a member due to forces acting upon it. The bending strength of material is the resistance it offers to deflecting forces. Torsion is a twisting force. Such action would occur in a member fixed at one end and twisted at the other. The torsional strength of material is its resistance to twisting.
The relationship between the strength of a material and its weight per cubic inch, expressed as a ratio, is known as the strength-to-weight ratio. This ratio forms the basis for comparing the desirability of various materials for use in airframe construction and repair. Neither strength nor weight alone can be used as a means of true comparison. In some applications, such as the skin of monocoque structures, thickness is more important than strength. In this instance, the material with the lightest weight for a given thickness or gauge is best. Thickness or bulk is necessary to prevent bucking or damage caused by careless handling.
Corrosion is the eating away or pitting of the surface or the internal structure of metals. Because of the thin sections and the safety factors used in aircraft design and construction, it would be dangerous to select a material possessing poor corrosion-resistant characteristics.
Another significant factor to consider in maintenance and repair is the ability of a material to be formed, bent, or machined to required shapes. The hardening of metals by cold working or forming is termed work hardening. If a piece of metal is formed (shaped or bent) while cold, it is said to be cold worked. Practically all the work an aviation mechanic does on metal is cold work. While this is convenient, it causes the metal to become harder and more brittle.
If the metal is cold worked too much, that is, if it is bent back and forth or hammered at the same place too often, it will crack or break. Usually, the more malleable and ductile a metal is, the more cold working it can stand. Any process that involves controlled heating and cooling of metals to develop certain desirable characteristics (such as hardness, softness, ductility, tensile strength, or refined grain structure) is called heat treatment or heat-treating. With steels, the term “heat-treating” has a broad meaning and includes processes such as annealing, normalizing, hardening, and tempering.
In the heat treatment of aluminum alloys, only two processes are included: the hardening and toughening process and the softening process. The hardening and toughening process is called heat-treating, and the softening process is called annealing. Aircraft metals are subjected to both shock and fatigue (vibrational) stresses. Fatigue occurs in materials that are exposed to frequent reversals of loading or repeatedly applied loads, if the fatigue limit is reached or exceeded. Repeated vibration or bending ultimately causes a minute crack to occur at the weakest point. As vibration or bending continues, the crack lengthens until the part completely fails. This is termed “shock and fatigue failure.” Resistance to this condition is known as shock and fatigue resistance. It is essential that materials used for critical parts be resistant to these stresses.
Heat treatment is a series of operations involving the heating and cooling of metals in the solid state. Its purpose is to change a mechanical property, or combination of mechanical properties, so that the metal is more useful, serviceable, and safe for a definite purpose. By heat-treating, a metal can be made harder, stronger, and more resistant to impact. Heat-treating can also make a metal softer and more ductile. No one heat-treating operation can produce all these characteristics. In fact, some properties are often improved at the expense of others. In being hardened, for example, a metal may become brittle.
The various heat-treating processes are similar in that they all involve the heating and cooling of metals. They differ, however, in the temperatures to which the metal is heated, the rate at which it is cooled, and, of course, in the result.
The most common forms of heat treatment for ferrous metals are hardening, tempering, normalizing, annealing, and casehardening. Most nonferrous metals can be annealed and many of them can be hardened by heat treatment. However, there is only one nonferrous metal, titanium, that can be casehardened, and none can be tempered or normalized.
An alloy may be in the form of a solid solution, a mechanical mixture, or a combination of a solid solution and a mechanical mixture. When an alloy is in the form of a solid solution, the elements and compounds that form the alloy are absorbed, one into the other, in much the same way that salt is dissolved in a glass of water, and the constituents cannot be identified even under a microscope.
When two or more elements or compounds are mixed but can be identified by microscopic examination, a mechanical mixture is formed. A mechanical mixture can be compared to the mixture of sand and gravel in concrete. The sand and gravel are both visible. Just as the sand and gravel are held together and kept in place by the matrix of cement, the other constituents of an alloy are embedded in the matrix formed by the base metal.
An alloy in the form of a mechanical mixture at ordinary temperatures may change to a solid solution when heated. When cooled back to normal temperature, the alloy may return to its original structure. On the other hand, it may remain a solid solution or form a combination of a solid solution and mechanical mixture. An alloy, which consists of a combination of solid solution and mechanical mixture at normal temperatures, may change to a solid solution when heated. When cooled, the alloy may remain a solid solution, return to its original structure, or form a complex solution.
In addition, using a furnace beyond its rated maximum temperature shortens its life and may necessitate costly and time-consuming repairs.
Fuel-fired furnaces (gas or oil) require air for proper combustion, and an air compressor or blower is therefore necessary. These furnaces are usually of the muffler type; that is, the combustion of the fuel takes place outside of and around the chamber in which the work is placed. If an open muffler is used, the furnace should be designed to prevent the direct impingement of flame on the work.
In furnaces heated by electricity, the heating elements are generally in the form of wire or ribbon. Good design requires incorporation of additional heating elements at locations where maximum heat loss may be expected. Such furnaces commonly operate at up to a maximum temperature of about 2,000 °F. Furnaces operating at temperatures up to about 2,500 °F usually employ resistor bars of sintered carbides.
Furnaces intended primarily for tempering may be heated by gas or electricity and are frequently equipped with a fan for circulating the hot air.
Salt baths are available for operating at either tempering or hardening temperatures. Depending on the composition of the salt bath, heating can be conducted at temperatures as low as 325 °F to as high as 2,450 °F. Lead baths can be used in the temperature range of 650 °F to 1,700 °F. The rate of heating in lead or salt baths is much faster in furnaces.
Heat-treating furnaces differ in size, shape, capacity, construction, operation, and control. They may be circular or rectangular and may rest on pedestals or directly on the floor. There are also pit-type furnaces, which are below the surface of the floor. When metal is to be heated in a bath of molten salt or lead, the furnace must contain a pot or crucible for the molten bath.
The size and capacity of a heat-treating furnace depends on the intended use. A furnace must be capable of heating rapidly and uniformly, regardless of the desired maximum temperature or the mass of the charge. An oven-type furnace should have a working space (hearth) about twice as long and three times as wide as any part that is heated in the furnace.
Accurate temperature measurement is essential to good heat-treating. The usual method is by means of thermocouples: the most common base metal couples are copper-constantan (up to about 700 °F), iron-constantan (up to about 1,400 °F), and chromel-alumel (up to about 2,200 °F). The most common noble metal couples (which can be used up to about 2,800 °F) are platinum coupled with either the alloy 87 percent platinum (13 percent rhodium) or the alloy 90 percent platinum (10 percent rhodium). The temperatures quoted are for continuous operation.
The life of thermocouples is affected by the maximum temperature (which may frequently exceed those given above) and by the furnace atmosphere. Iron-constantan is more suited for use in reducing and chromel-alumel in oxidizing atmospheres. Thermocouples are usually encased in metallic or ceramic tubes closed at the hot end to protect them from the furnace gases. A necessary attachment is an instrument, such as a millivoltmeter or potentiometer, for measuring the electromotive force generated by the thermocouple. In the interest of accurate control, place the hot junction of the thermocouple as close to the work as possible. The use of an automatic controller is valuable in controlling the temperature at the desired value.
Pyrometers may have meters either of the indicating type or recording type. Indicating pyrometers give direct reading of the furnace temperature. The recording type produces a permanent record of the temperature range throughout the heating operation by means of an inked stylus attached to an arm, which traces a line on a sheet of calibrated paper or temperature chart.
Pyrometer installations on all modern furnaces provide automatic regulation of the temperature at any desired setting. Instruments of this type are called controlling potentiometer pyrometers. They include a current regulator and an operating mechanism, such as a relay.
If temperature-measuring equipment is not available, it becomes necessary to estimate temperatures by some other means. An inexpensive, yet accurate method involves the use of commercial crayons, pellets, or paints that melt at various temperatures within the range of 125 °F to 1,600 °F.
The least accurate method of temperature estimation is by observation of the color of the hot hearth of the furnace or of the work. The heat colors observed are affected by many factors, such as the conditions of artificial or natural light, the character of the scale on the work, and so forth. Steel begins to appear dull red at about 1,000 °F, and as the temperature increases, the color changes gradually through various shades of red to orange, to yellow, and finally to white. A rough approximation of the correspondence between color and temperature is indicated in Figure 1.
Hot Working
Almost all steel is hot worked from the ingot into some form from which it is either hot or cold worked to the finished shape. When an ingot is stripped from its mold, its surface is solid, but the interior is still molten. The ingot is then placed in a soaking pit, which retards loss of heat, and the molten interior gradually solidifies. After soaking, the temperature is equalized throughout the ingot, then it is reduced to intermediate size by rolling, making it more readily handled.The rolled shape is called a bloom when its section dimensions are 6 inches × 6 inches or larger and square. The section is called a billet when it is square and less than 6 inches × 6 inches. Rectangular sections, which have a width greater than twice their thickness, are called slabs. The slab is the intermediate shape from which sheets are rolled.
Blooms, billets, or slabs are heated above the critical range and rolled into a variety of shapes of uniform cross section. Common rolled shapes are sheet, bar, channel, angle, and I-beam. Hot-rolled material is frequently finished by cold rolling or drawing to obtain accurate finish dimensions and a bright, smooth surface.
Complicated sections, which cannot be rolled, or sections of which only a small quantity is required, are usually forged. Forging of steel is a mechanical working at temperatures above the critical range to shape the metal as desired. Forging is done either by pressing or hammering the heated steel until the desired shape is obtained.
Pressing is used when the parts to be forged are large and heavy; this process also replaces hammering where high-grade steel is required. Since a press is slow acting, its force is uniformly transmitted to the center of the section, thus affecting the interior grain structure, as well as the exterior to give the best possible structure throughout.
Hammering can be used only on relatively small pieces. Since hammering transmits its force almost instantly, its effect is limited to a small depth. Thus, it is necessary to use a very heavy hammer or to subject the part to repeated blows to ensure complete working of the section. If the force applied is too weak to reach the center, the finished forged surface is concave. If the center was properly worked, the surface is convex or bulged. The advantage of hammering is that the operator has control over both the amount of pressure applied and the finishing temperature and can produce small parts of the highest grade. This type of forging is usually referred to as smith forging. It is used extensively where only a small number of parts are needed. Considerable machining time and material are saved when a part is smith forged to approximately the finished shape.
Steel is often harder than necessary and too brittle for most practical uses when put under severe internal strain. To relieve such strain and reduce brittleness, it is tempered after being hardened. This consists of heating the steel in a furnace to a specified temperature and then cooling it in air, oil, water, or a special solution. Temper condition refers to the condition of metal or metal alloys with respect to hardness or toughness. Rolling, hammering, or bending these alloys, or heat treating and aging them, causes them to become tougher and harder. At times, these alloys become too hard for forming and must be re-heat treated or annealed.
Metals are annealed to relieve internal stresses, soften the metal, make it more ductile, and refine the grain structure. Annealing consists of heating the metal to a prescribed temperature, holding it there for a specified length of time, and then cooling the metal back to room temperature. To produce maximum softness, the metal must be cooled very slowly. Some metals must be furnace cooled; others may be cooled in air.
Normalizing applies to iron base metals only. Normalizing consists of heating the part to the proper temperature, holding it at that temperature until it is uniformly heated, and then cooling it in still air. Normalizing is used to relieve stresses in metals.
Strength, weight, and reliability are three factors that determine the requirements to be met by any material used in airframe construction and repair. Airframes must be strong and yet as lightweight as possible. There are very definite limits to which increases in strength can be accompanied by increases in weight. An airframe so heavy that it could not support a few hundred pounds of additional weight would be of little use.
All metals, in addition to having a good strength-toweight ratio, must be thoroughly reliable, thus minimizing the possibility of dangerous and unexpected failures. In addition to these general properties, the material selected for a definite application must possess specific qualities suitable for the purpose.
The material must possess the strength required by the dimensions, weight, and use. The five basic stresses that metals may be required to withstand are tension, compression, shear, bending, and torsion.
The tensile strength of a material is its resistance to a force, which tends to pull it apart. Tensile strength is measured in pounds per square inch (psi) and is calculated by dividing the load in pounds required to pull the material apart by its cross-sectional area in square inches.
The compression strength of a material is its resistance to a crushing force, which is the opposite of tensile strength. Compression strength is also measured in psi. When a piece of metal is cut, the material is subjected, as it comes in contact with the cutting edge, to a force known as shear. Shear is the tendency on the part of parallel members to slide in opposite directions. It is like placing a cord or thread between the blades of a pair of scissors (shears). The shear strength is the shear force in psi at which a material fails. It is the load divided by the shear area.
Bending can be described as the deflection or curving of a member due to forces acting upon it. The bending strength of material is the resistance it offers to deflecting forces. Torsion is a twisting force. Such action would occur in a member fixed at one end and twisted at the other. The torsional strength of material is its resistance to twisting.
The relationship between the strength of a material and its weight per cubic inch, expressed as a ratio, is known as the strength-to-weight ratio. This ratio forms the basis for comparing the desirability of various materials for use in airframe construction and repair. Neither strength nor weight alone can be used as a means of true comparison. In some applications, such as the skin of monocoque structures, thickness is more important than strength. In this instance, the material with the lightest weight for a given thickness or gauge is best. Thickness or bulk is necessary to prevent bucking or damage caused by careless handling.
Corrosion is the eating away or pitting of the surface or the internal structure of metals. Because of the thin sections and the safety factors used in aircraft design and construction, it would be dangerous to select a material possessing poor corrosion-resistant characteristics.
Another significant factor to consider in maintenance and repair is the ability of a material to be formed, bent, or machined to required shapes. The hardening of metals by cold working or forming is termed work hardening. If a piece of metal is formed (shaped or bent) while cold, it is said to be cold worked. Practically all the work an aviation mechanic does on metal is cold work. While this is convenient, it causes the metal to become harder and more brittle.
If the metal is cold worked too much, that is, if it is bent back and forth or hammered at the same place too often, it will crack or break. Usually, the more malleable and ductile a metal is, the more cold working it can stand. Any process that involves controlled heating and cooling of metals to develop certain desirable characteristics (such as hardness, softness, ductility, tensile strength, or refined grain structure) is called heat treatment or heat-treating. With steels, the term “heat-treating” has a broad meaning and includes processes such as annealing, normalizing, hardening, and tempering.
In the heat treatment of aluminum alloys, only two processes are included: the hardening and toughening process and the softening process. The hardening and toughening process is called heat-treating, and the softening process is called annealing. Aircraft metals are subjected to both shock and fatigue (vibrational) stresses. Fatigue occurs in materials that are exposed to frequent reversals of loading or repeatedly applied loads, if the fatigue limit is reached or exceeded. Repeated vibration or bending ultimately causes a minute crack to occur at the weakest point. As vibration or bending continues, the crack lengthens until the part completely fails. This is termed “shock and fatigue failure.” Resistance to this condition is known as shock and fatigue resistance. It is essential that materials used for critical parts be resistant to these stresses.
Heat treatment is a series of operations involving the heating and cooling of metals in the solid state. Its purpose is to change a mechanical property, or combination of mechanical properties, so that the metal is more useful, serviceable, and safe for a definite purpose. By heat-treating, a metal can be made harder, stronger, and more resistant to impact. Heat-treating can also make a metal softer and more ductile. No one heat-treating operation can produce all these characteristics. In fact, some properties are often improved at the expense of others. In being hardened, for example, a metal may become brittle.
The various heat-treating processes are similar in that they all involve the heating and cooling of metals. They differ, however, in the temperatures to which the metal is heated, the rate at which it is cooled, and, of course, in the result.
The most common forms of heat treatment for ferrous metals are hardening, tempering, normalizing, annealing, and casehardening. Most nonferrous metals can be annealed and many of them can be hardened by heat treatment. However, there is only one nonferrous metal, titanium, that can be casehardened, and none can be tempered or normalized.
Internal Structure of Metals
The results obtained by heat treatment depend on the structure of the metal and on the way the structure changes when the metal is heated and cooled. A pure metal cannot be hardened by heat treatment, because there is little change in its structure when heated. On the other hand, most alloys respond to heat treatment since their structures change with heating and cooling.An alloy may be in the form of a solid solution, a mechanical mixture, or a combination of a solid solution and a mechanical mixture. When an alloy is in the form of a solid solution, the elements and compounds that form the alloy are absorbed, one into the other, in much the same way that salt is dissolved in a glass of water, and the constituents cannot be identified even under a microscope.
When two or more elements or compounds are mixed but can be identified by microscopic examination, a mechanical mixture is formed. A mechanical mixture can be compared to the mixture of sand and gravel in concrete. The sand and gravel are both visible. Just as the sand and gravel are held together and kept in place by the matrix of cement, the other constituents of an alloy are embedded in the matrix formed by the base metal.
An alloy in the form of a mechanical mixture at ordinary temperatures may change to a solid solution when heated. When cooled back to normal temperature, the alloy may return to its original structure. On the other hand, it may remain a solid solution or form a combination of a solid solution and mechanical mixture. An alloy, which consists of a combination of solid solution and mechanical mixture at normal temperatures, may change to a solid solution when heated. When cooled, the alloy may remain a solid solution, return to its original structure, or form a complex solution.
Heat-Treating Equipment
Successful heat treating requires close control over all factors affecting the heating and cooling of metals. Such control is possible only when the proper equipment is available and the equipment is selected to fit the job. Thus, the furnace must be of the proper size and type and must be controlled so that temperatures are kept within the limits prescribed for each operation. Even the atmosphere within the furnace affects the condition of the part being heat-treated. Further, the quenching equipment and the quenching medium must be selected to fit the metal and the heat-treating operation. Finally, there must be equipment for handling parts and materials, for cleaning metals, and for straightening parts.Furnaces and Salt Baths
There are many different types and sizes of furnaces used in heat treatment. As a rule, furnaces are designed to operate in certain specific temperature ranges and attempted use in other ranges frequently results in work of inferior quality.In addition, using a furnace beyond its rated maximum temperature shortens its life and may necessitate costly and time-consuming repairs.
Fuel-fired furnaces (gas or oil) require air for proper combustion, and an air compressor or blower is therefore necessary. These furnaces are usually of the muffler type; that is, the combustion of the fuel takes place outside of and around the chamber in which the work is placed. If an open muffler is used, the furnace should be designed to prevent the direct impingement of flame on the work.
In furnaces heated by electricity, the heating elements are generally in the form of wire or ribbon. Good design requires incorporation of additional heating elements at locations where maximum heat loss may be expected. Such furnaces commonly operate at up to a maximum temperature of about 2,000 °F. Furnaces operating at temperatures up to about 2,500 °F usually employ resistor bars of sintered carbides.
Temperature Measurement and Control
A thermoelectric instrument, known as a pyrometer, measures temperature in the heat-treating furnace. This instrument measures the electrical effect of a thermocouple and, hence, the temperature of the metal being treated. A complete pyrometer consists of three parts: a thermocouple, extension leads, and meter.Furnaces intended primarily for tempering may be heated by gas or electricity and are frequently equipped with a fan for circulating the hot air.
Salt baths are available for operating at either tempering or hardening temperatures. Depending on the composition of the salt bath, heating can be conducted at temperatures as low as 325 °F to as high as 2,450 °F. Lead baths can be used in the temperature range of 650 °F to 1,700 °F. The rate of heating in lead or salt baths is much faster in furnaces.
Heat-treating furnaces differ in size, shape, capacity, construction, operation, and control. They may be circular or rectangular and may rest on pedestals or directly on the floor. There are also pit-type furnaces, which are below the surface of the floor. When metal is to be heated in a bath of molten salt or lead, the furnace must contain a pot or crucible for the molten bath.
The size and capacity of a heat-treating furnace depends on the intended use. A furnace must be capable of heating rapidly and uniformly, regardless of the desired maximum temperature or the mass of the charge. An oven-type furnace should have a working space (hearth) about twice as long and three times as wide as any part that is heated in the furnace.
Accurate temperature measurement is essential to good heat-treating. The usual method is by means of thermocouples: the most common base metal couples are copper-constantan (up to about 700 °F), iron-constantan (up to about 1,400 °F), and chromel-alumel (up to about 2,200 °F). The most common noble metal couples (which can be used up to about 2,800 °F) are platinum coupled with either the alloy 87 percent platinum (13 percent rhodium) or the alloy 90 percent platinum (10 percent rhodium). The temperatures quoted are for continuous operation.
The life of thermocouples is affected by the maximum temperature (which may frequently exceed those given above) and by the furnace atmosphere. Iron-constantan is more suited for use in reducing and chromel-alumel in oxidizing atmospheres. Thermocouples are usually encased in metallic or ceramic tubes closed at the hot end to protect them from the furnace gases. A necessary attachment is an instrument, such as a millivoltmeter or potentiometer, for measuring the electromotive force generated by the thermocouple. In the interest of accurate control, place the hot junction of the thermocouple as close to the work as possible. The use of an automatic controller is valuable in controlling the temperature at the desired value.
Pyrometers may have meters either of the indicating type or recording type. Indicating pyrometers give direct reading of the furnace temperature. The recording type produces a permanent record of the temperature range throughout the heating operation by means of an inked stylus attached to an arm, which traces a line on a sheet of calibrated paper or temperature chart.
Pyrometer installations on all modern furnaces provide automatic regulation of the temperature at any desired setting. Instruments of this type are called controlling potentiometer pyrometers. They include a current regulator and an operating mechanism, such as a relay.
Heating
The object in heating is to transform pearlite (a mixture of alternate strips of ferrite and iron carbide in a single grain) to austenite as the steel is heated through the critical range. Since this transition takes time, a relatively slow rate of heating must be used. Ordinarily, the cold steel is inserted when the temperature in the furnace is from 300 °F to 500 °F below the hardening temperature. In this way, too rapid heating through the critical range is prevented.If temperature-measuring equipment is not available, it becomes necessary to estimate temperatures by some other means. An inexpensive, yet accurate method involves the use of commercial crayons, pellets, or paints that melt at various temperatures within the range of 125 °F to 1,600 °F.
The least accurate method of temperature estimation is by observation of the color of the hot hearth of the furnace or of the work. The heat colors observed are affected by many factors, such as the conditions of artificial or natural light, the character of the scale on the work, and so forth. Steel begins to appear dull red at about 1,000 °F, and as the temperature increases, the color changes gradually through various shades of red to orange, to yellow, and finally to white. A rough approximation of the correspondence between color and temperature is indicated in Figure 1.
 |
| Figure 1. Temperature chart indicating conversion of Centigrade to Fahrenheit or vice versa, color temperature scale for hardening temperature range, and tempering temperature range |
It is also possible to secure some idea of the temperature of a piece of carbon or low alloy steel, in the low temperature range used for tempering, from the color of the thin oxide film that forms on the cleaned surface of the steel when heated in this range. The approximate temperature/color relationship is indicated on the lower portion of the scale in Figure 1.
It is often necessary or desirable to protect steel or cast iron from surface oxidation (scaling) and loss of
carbon from the surface layers (decarburization). Commercial furnaces, therefore, are generally equipped with some means of atmosphere control. This usually is in the form of a burner for burning controlled amounts of gas and air and directing the products of combustion into the furnace muffle. Water vapor, a product of this combustion, is detrimental and many furnaces are equipped with a means for eliminating it. For furnaces not equipped with atmosphere control, a variety of external atmosphere generators are available. The gas so generated is piped into the furnace and one generator may supply several furnaces. If no method of atmosphere control is available, some degree of protection may be secured by covering the work with cast iron borings or chips. Since the liquid heating medium surrounds the work in salt or lead baths, the problem of preventing scaling or decarburization is simplified. Vacuum furnaces also are used for annealing steels, especially when a bright non-oxidized surface is a prime consideration.
There are three commonly used quenching liquids: brine, water, and oil. Brine is the strongest quenching medium, water is next, and oil is the least. Generally, an oil quench is used for alloy steels and brine or water for carbon steels.
Brine usually is made of a 5 to 10 percent solution of salt (sodium chloride) in water. In addition to its greater cooling speed, brine has the ability to “throw” the scale from steel during quenching. Their temperature considerably affects the cooling ability of both water and brine, particularly water. Both should be kept cold—well below 60 °F. If the volume of steel being quenched tends to raise the temperature of the bath appreciably, add ice or use some means of refrigeration to cool the quenching bath.
There are many specially prepared quenching oils on the market; their cooling rates do not vary widely. A straight mineral oil with a Saybolt viscosity of about 100 at 100 °F is generally used. Unlike brine and water, the oils have the greatest cooling velocity at a slightly elevated temperature—about 100–140 °F—because of their decreased viscosity at these temperatures.
When steel is quenched, the liquid in immediate contact with the hot surface vaporizes; this vapor reduces the rate of heat abstraction markedly. Vigorous agitation of the steel or the use of a pressure spray quench is necessary to dislodge these vapor films and thus permit the desired rate of cooling.
The tendency of steel to warp and crack during the quenching process is difficult to overcome because certain parts of the article cool more rapidly than others. The following recommendations greatly reduce the warping tendency.
Tank location in reference to the heat-treating furnace is very important. Situate the tank to permit rapid transfer of the part from the furnace to the quenching medium. A delay of more than a few seconds, in many instances, proves detrimental to the effectiveness of the heat treatment. During transfer to the quench tank, employ guard sheets to retard the loss of heat when heat treating material of thin section. Provide a rinse tank to remove all salt from the material after quenching if the salt is not adequately removed in the quenching tank.
It is often necessary or desirable to protect steel or cast iron from surface oxidation (scaling) and loss of
carbon from the surface layers (decarburization). Commercial furnaces, therefore, are generally equipped with some means of atmosphere control. This usually is in the form of a burner for burning controlled amounts of gas and air and directing the products of combustion into the furnace muffle. Water vapor, a product of this combustion, is detrimental and many furnaces are equipped with a means for eliminating it. For furnaces not equipped with atmosphere control, a variety of external atmosphere generators are available. The gas so generated is piped into the furnace and one generator may supply several furnaces. If no method of atmosphere control is available, some degree of protection may be secured by covering the work with cast iron borings or chips. Since the liquid heating medium surrounds the work in salt or lead baths, the problem of preventing scaling or decarburization is simplified. Vacuum furnaces also are used for annealing steels, especially when a bright non-oxidized surface is a prime consideration.
Soaking
The temperature of the furnace must be held constant during the soaking period, since it is during this period that rearrangement of the internal structure of the steel takes place. Soaking temperatures for various types of steel are specified in ranges varying as much as 100 °F. [Figure 2] Small parts are soaked in the lower part of the specified range and heavy parts in the upper part of the specified range. The length of the soaking period depends upon the type of steel and the size of the part. Naturally, heavier parts require longer soaking to ensure equal heating throughout. As a general rule, a soaking period of 30 minutes to 1 hour is sufficient for the average heat-treating operation.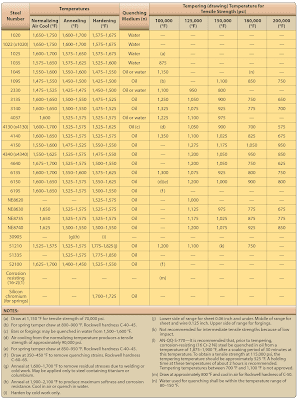 |
| Figure 2. Heat treatment procedures for steels |
Cooling
The rate of cooling through the critical range determines the form that the steel retains. Various rates of cooling are used to produce the desired results. Still air is a slow cooling medium but is much faster than furnace cooling. Liquids are the fastest cooling media and are therefore used in hardening steels.There are three commonly used quenching liquids: brine, water, and oil. Brine is the strongest quenching medium, water is next, and oil is the least. Generally, an oil quench is used for alloy steels and brine or water for carbon steels.
Quenching Media
Quenching solutions act only through their ability to cool the steel. They have no beneficial chemical action on the quenched steel and in themselves impart no unusual properties. Most requirements for quenching media are met satisfactorily by water or aqueous solutions of inorganic salts, such as table salt or caustic soda, or by some type of oil. The rate of cooling is relatively rapid during quenching in brine, somewhat less rapid in water, and slow in oil.Brine usually is made of a 5 to 10 percent solution of salt (sodium chloride) in water. In addition to its greater cooling speed, brine has the ability to “throw” the scale from steel during quenching. Their temperature considerably affects the cooling ability of both water and brine, particularly water. Both should be kept cold—well below 60 °F. If the volume of steel being quenched tends to raise the temperature of the bath appreciably, add ice or use some means of refrigeration to cool the quenching bath.
There are many specially prepared quenching oils on the market; their cooling rates do not vary widely. A straight mineral oil with a Saybolt viscosity of about 100 at 100 °F is generally used. Unlike brine and water, the oils have the greatest cooling velocity at a slightly elevated temperature—about 100–140 °F—because of their decreased viscosity at these temperatures.
When steel is quenched, the liquid in immediate contact with the hot surface vaporizes; this vapor reduces the rate of heat abstraction markedly. Vigorous agitation of the steel or the use of a pressure spray quench is necessary to dislodge these vapor films and thus permit the desired rate of cooling.
The tendency of steel to warp and crack during the quenching process is difficult to overcome because certain parts of the article cool more rapidly than others. The following recommendations greatly reduce the warping tendency.
- Never throw a part into the quenching bath. By permitting it to lie on the bottom of the bath, it is apt to cool faster on the topside than on the bottom side, thus causing it to warp or crack.
- Agitate the part slightly to destroy the coating of vapor that could prevent it from cooling evenly and rapidly. This allows the bath to dissipate its heat to the atmosphere.
- Immerse irregular shaped parts so that the heavy end enters the bath first.
Quenching Equipment
The quenching tank should be of the proper size to handle the material being quenched. Use circulating pumps and coolers to maintain approximately constant temperatures when doing a large amount of quenching. To avoid building up a high concentration of salt in the quenching tank, make provisions for adding fresh water to the quench tank used for molten salt baths.Tank location in reference to the heat-treating furnace is very important. Situate the tank to permit rapid transfer of the part from the furnace to the quenching medium. A delay of more than a few seconds, in many instances, proves detrimental to the effectiveness of the heat treatment. During transfer to the quench tank, employ guard sheets to retard the loss of heat when heat treating material of thin section. Provide a rinse tank to remove all salt from the material after quenching if the salt is not adequately removed in the quenching tank.
